Cartography: Andy

In the pharmacy of the Second Affiliated Hospital of Nanchang University, pharmacists are distributing purchased drugs to patients. People’s vision

A hospital in Wuhan publicized the centralized drug purchase policy to patients. Photo by Xin Ming Sun
Recently, a number of drugs have been reduced in price, including metformin, captopril, valsartan and other drugs. On August 24th, the results of the third batch of centralized drug procurement organized by the state (hereinafter referred to as centralized drug procurement), which is the largest to date, were announced. A total of 191 brand products of 55 drugs were selected, with an average price reduction of 53%.
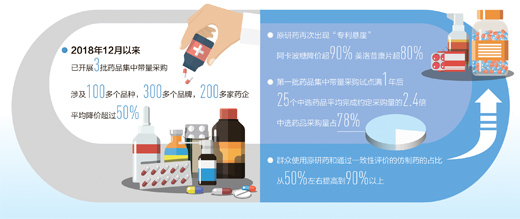
Since December 2018, the state organized centralized drug procurement in 11 cities including Beijing and Tianjin (hereinafter referred to as the "4+7" pilot), the number of purchased drugs has reached more than 100, and the number of products with different brands has exceeded 300, involving more than 200 selected enterprises. To the surprise of the vast number of insured patients, every collection has greatly reduced the price of some high-priced but large-dosage good drugs and big-name drugs, with an average reduction of more than half, which greatly reduced the burden of medication, such as gefitinib (Iressa), acarbose (Betanping), and entecavir, the first-line drug for patients with lung cancer, and the first-line drug for patients with diabetes.
Third-party evaluation shows that the quality and efficacy of the selected products have been generally recognized and accepted by doctors and patients. For the pharmaceutical industry, although it has experienced pain, it has improved the industry ecology and promoted benign development. In the future, more kinds of drugs will enter centralized procurement, which will better protect people’s demand for drugs and boost the construction of healthy China.
The price of each batch of centralized drugs is reduced by more than half.
The centralized procurement of drugs organized by the state brings huge consumption, and with the great advantage of super group purchase, it is exchanged for high quality and low price of drugs.
Uncle Hong, 76, from Xiamen City, Fujian Province, has been suffering from diabetes for more than 20 years and hypertension at the same time. More than 10 years ago, Uncle Hong began to take Baitangping, which costs 3 boxes a month, each box costs more than 90 yuan, and it costs about 3,300 yuan a year, which is a bit heavy. Recently, Uncle Hong went to the hospital to prescribe medicine and found that the medicine was cheap, and it only cost about 330 yuan a year, which is 1/10 of the original cost. "I asked the doctor if he prescribed the wrong medicine. I couldn’t believe it. Thanks to the country’s good policies, it has saved money for our diabetic patients and can also use big-name drugs. " Uncle Hong said.
Not only patients with chronic diseases, but also some seriously ill patients are greatly relieved.
Mr. Chen from Tieling City, Liaoning Province is a patient with bone metastasis from lung cancer. He has been taking gefitinib in recent years. In the first half of 2019, when he saw that the box of gefitinib in Shenyang dropped to 547 yuan, which was more than 1,700 yuan cheaper than that in Tieling, he asked someone to buy medicine in Shenyang. At the end of 2019, Tieling City also reduced its price. He calculated that according to the consumption of 3 boxes per month, he would be reimbursed by 56%, which would cost less than 5,000 yuan per month and reduce the annual drug cost by 60,000 yuan.
In August this year, Mr. Wu, a resident of Shanghai, went to Tongren Hospital affiliated to Shanghai Jiaotong University School of Medicine and prescribed a box of abiraterone acetate tablets for the treatment of malignant tumors as usual. He was pleasantly surprised to find that the drug with the original price of 16,268 yuan is now only 2,800 yuan, and the price reduction is over 80%. After the medical insurance reimbursement, the personal burden is 1,100 yuan.
This is the reform dividend brought by the centralized procurement of drugs organized by the state, benefiting countless families of patients. Since December 2018, after the pilot, the state has organized all localities to form alliances with provinces as units, and entrusted the joint procurement office to carry out centralized and quantitative procurement of cross-regional alliances. Each region collects the consumption of drugs in local public hospitals, and the selected drugs will be given 50% to 80% of the market consumption, and the medical insurance will pay in advance. In the case of consistency evaluation and quality certification, the centralized drug procurement organized by the state brings huge consumption, and with the great advantage of super group purchase, it gets high quality and low price of products.
The data show that the selected drugs are popular in clinic, and the completion degree of agreed dosage is high. After the expiration of the "4+7" pilot project for one year, the average purchase amount of 25 selected drugs was 2.4 times, and the purchase amount of selected drugs accounted for 78% of the purchase amount of the same drug.
Shanghai is one of the "4+7" pilot cities, and it is also the earliest area to purchase with quantity. Li Ling is the chief pharmacist in the pharmacy department of Tongren Hospital affiliated to Shanghai Jiaotong University School of Medicine, and also the vice chairman of the hospital pharmacy professional committee of Shanghai Pharmaceutical Association. She told reporters that Shanghai has done a good job in implementing the supporting policies of national procurement with quantity, such as leaving "air holes", and you can choose drugs from the middle or non-middle. Hospital publicity is well done and implemented, even if the original drug is replaced by generic drug, the patient’s acceptance is relatively high. Shanghai also uses near infrared spectroscopy to detect each batch of drugs to ensure excellent quality. Since the implementation, there are only a few reports of clinical adverse reactions, which are highly recognized.
No longer "only low prices are taken"
The entry threshold of high quality requirements means that the quality of the selected drugs is guaranteed even if the price is low.
Amlodipine besylate tablets of 0.15 yuan each, paracetamol tablets of 0.034 yuan each, captopril tablets of 0.014 yuan each, metformin hydrochloride tablets of 0.015 yuan each … … The price of some selected drugs is as low as 10 cents and 1 cent. Is the quality reliable? Is this kind of centralized purchasing going back to the old road of "only taking low prices"?
Gong Bo, director of the Price Mining Department of Shanghai Medical Insurance Bureau, said that in the "4+7" pilot, there were not many generic drug companies that passed the consistency evaluation, and the scope of the procurement pilot was not large. In order to reflect the scale effect, only the drugs with the lowest quotation were eligible for the selection. However, from the beginning of the national expansion of centralized procurement, the variety specifications increased, and the procurement volume expanded to the whole country. According to the market competition pattern, the maximum number of enterprises that can be selected from the middle was determined. In the third batch of centralized procurement, the maximum number of enterprises that can be selected from a variety was expanded to eight. The drugs participating in the centralized collection are generic drugs that have passed the consistency evaluation, and the quality level is high. In this case, the winning bid is different from the previous "only low price is taken" that does not distinguish the quality level. The entry threshold of high quality requirements also means that even if the price of the selected drug is low, the quality is guaranteed.
The reporter interviewed some patients who originally used the original drug and later switched to the generic drug. In the general clinic of Wuliqiao Community Health Service Center in Shanghai, Uncle Lin, who has been taking Bai Tangping for more than 10 years, is prescribing medicine. Last month, Uncle Lin changed the drugs selected from the centralized collection. "At first, I was afraid that the effect would not be good. I opened a box first. I tried it well and chose it." Uncle Lin said.
Huang Lei, director of Wuliqiao Community Health Service Center, said that there are currently 26 kinds of national centralized drugs used in hospitals, and amlodipine besylate and acarbose are commonly used drugs for patients with hypertension and diabetes respectively. Although they are no longer original drugs, more than 200 boxes can be prescribed every day. "These drugs have a large dosage and a large decline. After dressing change, the effect is good and they are welcomed by patients." April 20 this year was the first day of the second batch of centralized drugs, and many patients went to the hospital to buy drugs that day.
For some drugs that have dropped to "ultra-low price", experts have analyzed that some small enterprises are using "ultra-low price" to win the chance of winning because they will die if they miss, and they will survive if they win, but it is still difficult to survive if there are no new varieties to follow up. In contrast, large enterprises are becoming more and more rational, their costs are well controlled, their quality is guaranteed, and their product lines are stable. With the optimization of market formats, they will win from them and win the market.
It has been suggested that at such a low price, the profit margin of enterprises is too small, which will affect innovation. A number of companies said that generic drugs do not involve research and development costs, so the cost is relatively low. In many countries, generic drugs are small profits but quick turnover. Enterprises have their own strategic considerations whether to reduce prices for the market.
For enterprises, generic drugs and innovative drugs are two systems, and it is impossible to earn back the money for developing innovative drugs by selling generic drugs. From the international experience, the financing of innovative drugs is rarely filled by the profits of generic drugs, but more by capital markets such as venture capital and stock market.
In the third batch of centralized procurement, Jiangsu Haosen Pharmaceutical Co., Ltd. has 6 products to participate in the bidding and 5 products were selected. In the three batches of centralized purchasing, several products of Hausen were selected at a large price reduction, but it did not affect the pace of innovation. The relevant person in charge of the company said that the centralized procurement rules are becoming more and more perfect, the hospital implementation is in place, and the actual consumption is far greater than the reported amount, which gives the enterprise great confidence. Enterprises are actively transforming into innovative enterprises. At present, there are several 1.1 innovative drugs waiting to be listed in the fields of hypoglycemic, tumor and hematology.
The "patent cliff" appeared
Centralized purchasing with quantity makes it impossible to maintain the high price of the original research drugs that have passed the patent period, and the price has dropped sharply, so that patients can afford big-name drugs.
In these rounds of centralized procurement, some high-priced original research drugs have encountered "patent cliff" one after another, and some people called for the original research drugs to "retreat" in centralized procurement. However, this is not the case.
The original drug "patent cliff" first appeared in the "4+7" pilot in December 2018. At that time, the original research drug gefitinib reported the lowest price in the world of 547 yuan, which was 76% lower than the original price. In the industry’s view, this is not only a price reduction, but also a new drug price era of the original research drug "patent cliff".
The "patent cliff" refers to the phenomenon that the price of generic drugs has dropped sharply after the patent of the original drug expires. In China, after many patented drugs expired, generic drugs failed to appear. Even if it appears, because there is no conformity evaluation quality certification, it is impossible to confirm the quality level of generic drugs at the time of bidding, and the procurement with quantity has not been really implemented. Therefore, the original research drugs that have passed the patent period still maintain high drug prices and occupy a large market share. Generic drugs can’t replace the original drugs, and the drug burden of patients remains high.
At the time of the "4+7" pilot, the situation has completely changed. The third generation of targeted drugs for non-small cell lung cancer has been listed, and the first imitation drug in China has also been listed. As the first generation product, gefitinib will soon be eliminated from the market. In the case that a large number of generic drugs have passed the consistency evaluation and the country began to organize centralized procurement of drugs with quantity, the price of gefitinib, the original research drug, has become a wise choice to cope with market competition and has become the first patent cliff in the history of Chinese medicine.
In the second batch of drug collection in January this year, the original drug "patent cliff" appeared again. The price reduction of acarbose, the original drug for diabetes, exceeded 90%; Meloxicam tablets, the original research drug for the treatment of rheumatoid arthritis, reduced its price by more than 80%, and the price per tablet dropped from 2 yuan to 0.18 yuan.
In the third batch of centralized drug procurement organized by the state, more original drugs were involved. Although only three original drugs were selected, as in the previous collections, the original drug research enterprises actively participated, not "retreated".
Gong Bo said: "The price of original drugs is generally high, and the price is changed by quantity, which requires a sharp price reduction of drugs. In drug collection at home and abroad, the selection of generic drugs is a normal phenomenon, and the selection of original drugs is an exception. " From the point of view of the original drug research enterprise, the failure to choose is closely related to the development strategy of the enterprise itself. Whether to occupy the selected market or the non-selected market, what impact it will have on major domestic and foreign markets, etc., all need to be considered.
Hu Shanlian, a professor at the School of Public Health of Fudan University, said that the pharmaceutical companies that can significantly reduce prices are mainly due to the high cost of circulation. Most of these successful bidders control the source of raw materials, so they can achieve significant price cuts. Some of the original drugs have been greatly reduced in price in order that a new generation of products can quickly occupy the market. "This shows the direction of innovation in the future and will bring a new atmosphere to the entire pharmaceutical industry."
The data shows that from the situation of "4+7" pilot areas, the proportion of people using original drugs and generic drugs that passed the consistency evaluation has greatly increased from about 50% to over 90%, and the quality level of patients’ medication has improved significantly.
"Selling with gold" disappeared.
Centralized procurement with quantity promotes the replacement of high-priced original research drugs with cheap generic drugs, and the low-priced drugs that patients love are revived. Enterprises pay attention to product quality and no longer promote sales with kickbacks.
Centralized purchasing with quantity not only promotes generic drugs to replace original drugs, but also forces enterprises to change the unreasonable "sales with gold" model and guide enterprises to pay attention to product quality instead of focusing on sales, thus changing the industry ecology.
In the past, some drugs were sold with money. Drugs are given to agents at the reserve price, and they are sold by layers of rebates. The price can often be several times or even ten times higher than the ex-factory price, resulting in a false high price.
"In the past, there was no quantity in centralized bidding for drugs. Although the drugs won the bid, there was still a threshold for entering the hospital, and ‘ Sales with gold ’ Mode. Centralized purchasing with quantity allows enterprises to have expectations, understand that there will be a market if they are selected, and can comprehensively consider factors such as cost, market and development strategy to quote. Say goodbye to the unreasonable ‘ Sales with gold ’ The model will help improve the industry ecology and promote the sound development of the industry. " Wu Ming, assistant director of Peking University School of Medicine and professor of health economics, said.
Due to the change of "sales with gold" mode, in the second batch of centralized purchase, some low-priced drugs that patients used to love were revived and returned to the market. For example, acetaminophen, an antipyretic and analgesic drug, was selected at a price of 0.03 to 0.07 yuan/tablet, slightly higher than the lowest selling price of 0.02 yuan/tablet in history, which made low-priced drugs have a certain profit and gained a market, as well as metronidazole for toothache and amoxicillin for standing antibiotics.
These low-priced drugs have low production costs and fierce competition. In the past, due to the circulation mode, it was impossible to afford the cost of "selling with gold", but it was difficult for low-priced drugs to open the market and was "reversely eliminated" by high-priced drugs, making it difficult for patients to buy drugs at low prices. This is the well-known phenomenon of drug "price reduction to death" in the past. After the centralized procurement and use of drugs organized by the state, enterprises no longer need to carry out sales public relations through purchasing with quantity and ensuring use, and low-priced drugs can be "resurrected" and returned to the market.
Some quality-oriented enterprises win the market through centralized procurement. In the third batch of centralized purchase, Azacytidine for injection of Sichuan Huiyu Pharmaceutical was selected from each 260 yuan. It is the first imitation drug, which has been listed in Europe for the first time before. Through centralized purchasing, this product can enter the hospital without setting up a sales team to realize small profits but quick turnover. "We pay attention to product quality, and the products quickly pass the consistency evaluation when they come out. Now you can enter the hospital through centralized procurement, which is also the signal that quantity procurement sends to the market — — Excellent quality can enter the hospital and win the market. " The person in charge of Sichuan Huiyu Pharmaceutical said.
Centralized purchasing is also beneficial to enterprises with listing licenses. Listed license holders (enterprises) who focus on product research and development have no sales team. The policy of purchasing with quantity makes this kind of products directly enter the hospital, without the extra cost of "selling with gold".
Shanghai Anbisheng Pharmaceutical Co., Ltd. is a listed license holder enterprise. During the "4+7" pilot project, Ji Ran, the general manager of the company, thought that the cost of academic meetings and marketing promotion could be reduced to ensure profits. In the third batch of centralized purchasing, the enterprise products were selected again.
Wu Ming believes that from the current situation, centralized drug collection is a breakthrough in the reform of compensation mechanism in public hospitals and the promotion of "three medical linkages". After several rounds of centralized procurement, the number of selected drugs increased, and the amount of money accounted for a higher and higher proportion of medical insurance payment, which was gradually recognized by doctors and patients. At the same time, due to the sharp drop in prices, medical insurance frees up space to optimize the drug structure, which is conducive to drug innovation. The surplus hospitals that purchase drugs from medical insurance can be retained, which urges hospitals to actively control costs and incite the reform of public hospitals.
More drugs usher in "super group purchase"
Centralized purchasing with quantity enters the normal operation track, which means that patients can use high-quality and high-priced drugs for a long time, benefiting more patients.
Since the second batch of centralized procurement, the state has organized drug centralized procurement to enter a normal operation track. In the future, more and more products will be included in centralized procurement with quantity.
From July 15th to 16th this year, the National Medical Insurance Bureau held a forum to listen to experts’ opinions and suggestions on centralized procurement of biological products (including insulin) and Chinese patent medicines, study and improve procurement policies in related fields, and promote the reform of procurement methods. This releases the signal that more drugs will be brought into centralized collection.
Normalization of centralized purchasing means that high-quality and high-priced drugs can be used for a long time, not only western medicines, but also Chinese patent medicines and insulin injections commonly used by diabetic patients in the future. Shi Luwen, a professor at Peking University School of Pharmacy, said: "The centralized drug collection mechanism organized by the state is closely linked with its procurement, payment, payment and use, ‘ Sanyi ’ The coordination of linkage ensures the quality of drugs and timely supply. For patients, they can enjoy long-term reform dividends and reduce drug costs. At the same time, because the overall quality of the selected drugs is good, gradually establishing recognition of the national pharmaceutical industry can better safeguard people’s health. "
At present, there are only 112 kinds of drugs involved in the three batches of centralized purchase, but the basic drugs for urban workers’ medical insurance in China have reached more than 2,000 kinds, and the number of drugs that have not entered the centralized purchase and failed the conformity evaluation of generic drugs is still large. However, under the effect of centralized production with quantity, more and more drugs are facing the pressure of price reduction.
According to the relevant regulations, the payment standard will be gradually adjusted for some drugs whose prices are quite different from those of the selected drugs, and will be adjusted in place within two or three years; In some places, special centralized collection with quantity is carried out for some drug varieties. In August this year, Shanghai issued a document to encourage public medical institutions in this city to explore and carry out centralized bargaining procurement for drugs selected from non-centralized procurement in the form of medical consortia, monomers or voluntary procurement alliances.
In the future, the mechanism of centralized mining with quantity still needs to be improved in practice. In the interview, some pharmaceutical companies told reporters that some drugs have been collected and their prices are close to the "floor price". It is suggested that the same medical insurance payment standard as the collection price should be enabled, so that China pharmaceutical enterprises can have more development space and patients can choose more space; According to the rules of centralized procurement, three companies of the same drug can automatically trigger centralized procurement through consistency evaluation, but some products have not been evaluated for two or three years after consistency evaluation, so they cannot participate in centralized procurement; Some hospitals expressed the hope to implement the method of "surplus retention and reasonable cost overruns sharing" as soon as possible to improve the enthusiasm of medical institutions to adopt selected drugs.
"With the expansion of centralized procurement coverage, the continuous improvement of rules and the coordinated efforts of supporting policies, the effect of purchasing with quantity will become more and more prominent, and the four effects at the beginning of policy design will be gradually realized, namely, drug price reduction and quality assurance, transformation and upgrading of the pharmaceutical industry, deepening reform of public hospitals, and reducing the burden and increasing efficiency of medical security." Wu Ming said.